Cryo-EM structure of yeast telomerase provides unprecedented insights into structure and function of telomerase holoenzymes and highlights diversity across eukaryotes
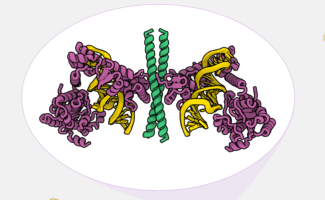
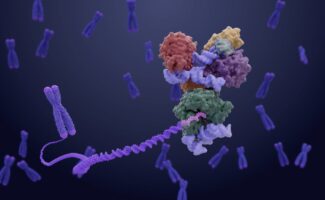
Telomeres are protective nucleoprotein complexes which cap the ends of chromosomes to preserve genome stability. Though they are gradually shortened during cell division, the holoenzyme complex telomerase works to offset this by adding telomeric DNA repeats to chromosome ends. The work of Kelly Nguyen’s group, in the LMB’s Structural Studies Division, alongside other researchers from around the world has highlighted the structural diversity of telomerase in different organisms. One model organism, the budding yeast Saccharomyces cerevisiae, has been integral to telomere research for decades. However, until now, no structural information existed due to its complex composition. Addressing this, Kelly’s group has used electron cryomicroscopy (cryo-EM) to determine the first structure of yeast telomerase, revealing significant divergences from other eukaryotic telomerase structures.
S. cerevisiae telomerase is difficult to biochemically and structurally study as it is far more complex than its human counterpart. It boasts 1157 RNA nucleotides, compared to just 451 in human telomerase. Additionally, it is a low abundance protein complex, meaning it requires additional enrichment procedures to generate enough sample to study.
To address the latter problem, Hongmiao Hu and Elsa Franco Echevarria, Investigator Scientists in Kelly’s group, worked with the LMB’s Media Prep facility to establish a strategy to make minute amounts of yeast telomerase from 50 litre batches of yeast culture. With the sample prepared, Hongmiao then worked with the LMB’s Electron Microscopy facility to generate the cryo-EM structures. Their work was further complemented by biochemical analyses on yeast strains by Hannah Neumann in Pascal Chartrand’s group at Université de Montreal and Gabriela Teplitz in Raymund Wellinger’s group at Université de Sherbrooke.
The resulting S. cerevisiae telomerase structure reveals that several distant regions of its RNA component, TLC1, converge together to form a compact core alongside three ever shorter telomere (Est) proteins and a Pop1/6/7 heterotrimer complex. Long RNA linkers are also present to flexibly tether a yKu70/80 heterodimer and a Sm heptamer.
In the new structure, the team discovered that the Pop 1/6/7 sub-complex is a component that is uniquely vital to telomere maintenance. The complex is shared with RNase P/MRP, involved in RNA processing. But, crucially, it also facilitates the association of Est1-3 making it key for telomerase assembly. Interestingly, the group found that the Pop1 loop was necessary for telomere maintenance but dispensable for RNA processing, pointing to the complex’s specific role in telomere maintenance.
Additionally, the structure illuminates how the Est protein, Est3, acts as a ‘molecular glue’ to stabilise holoenzyme assembly. When the group disrupted Est3 interactions with Est2 and Pop1, telomeres began to shorten, and senescence, the biological process of gradual deterioration with age, accelerated. Structural analysis found that Est3 is a homolog of the shelterin component TPP1 in human telomerase. Previous work from Kelly’s group identified that TPP1 is crucial to the recruitment of telomeres to telomerase in humans and for the regulation of telomerase activity during DNA replication.
Finally, diverting from other existing telomerase structures, the team identified a zinc finger motif (ZnF) within the yeast telomerase catalytic subunit. This ZnF was found to be essential for telomerase activity both in vitro and in vivo, likely as a stabiliser for the RNA template during catalysis. This zinc finger motif is also conserved in the yeasts Saccharomycetales and Saccharomycodales.
This work provides unprecedented insights into the structure and function of telomerase holoenzymes that has taken over 30 years to reconcile and highlights the evolutionary diversity of the complex across eukaryotic organisms. Additionally, the work holds potential clinical importance as dysregulation of telomerase activity is closely linked with cancers and premature ageing disorders. Therefore, a better understanding of the structure, mechanisms and regulation of telomerase has significant implications for human health. Moreover, understanding the diverse molecular assembly of telomerase in different species provides opportunities to develop novel interventionist therapeutics to target the proliferation and spread of pathogenic eukaryotes.
This work was funded by UKRI MRC, EMBO, Canadian Institutes of Health Research (CIHR), Canada Research Chairs and the Centre for Research in Aging.
Further references
Kelly’s group page
Raymund Wellinger – Université de Sherbrooke
Pascal Chartrand – Université de Montreal
Electron Microscopy Facility
Media Prep & Glasswash Facility
Related articles
First structure of human telomerase in dimeric assembly
Cryo-EM structures reveal the unexpected ability of shelterin to modulate telomeric nucleosomes
Cryo-EM structures reveal molecular basis of human telomerase recruitment
First atomic model of human telomerase constructed by electron cryo‑microscopy