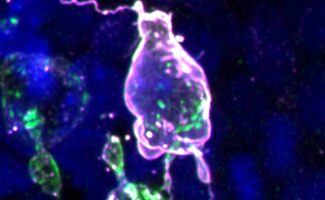
Mammalian primordial germ cells use actin-rich protrusions to navigate restrictive spaces during their developmental journey that increases their inherent risk to DNA damage
Primordial germ cells (PGCs) are unique in developing embryos as the only cells which carry the genetic material for future offspring. Ultimately becoming germ cells, either sperm or eggs, PGCs arise early in embryogenesis and often need to travel great distances to the site of the future gonads. Developmental biologists have found that different species employ distinct mechanisms to facilitate PGC migration, but little is known about how this works in mammals. For the first time, Katharine Goodwin and Kate McDole, based in the LMB’s Cell Biology Division, have imaged PGC migration in the developing mouse embryo, revealing how the cells use protrusions to navigate tight intercellular spaces and the surprising genetic damage they sustain during their journey.
PGCs in mouse embryos are first identifiable around 6.5 days after conception. Across three to five days, they follow external chemotactic signals to migrate through developing tissues to reach the site of the future gonads. At this early stage, the developing embryo is incredibly fragile so there are several challenges to its in-depth observation. To combat this, Kate and Katharine employed an interdisciplinary approach, key to which was a bespoke light sheet microscope and advanced microscopy methods, which allowed them to visualize PGC migration with high-spatiotemporal resolution over a three-day time period.
Assisted by collaborators Theresa Anne Emrich and Sebastian Arnold at the University of Freiburg, Germany, the group discovered that mouse PGC migration strategy is indeed distinct from non-mammalian species. Notably, mouse PGCs extend dynamic, actin-rich protrusions to aid navigation through extracellular matrix (ECM) barriers and tight intercellular spaces. PGCs also produce their own ECM proteins providing a possible scaffold on which they can migrate, suggesting that they remodel their immediate microenvironment to assist movement.
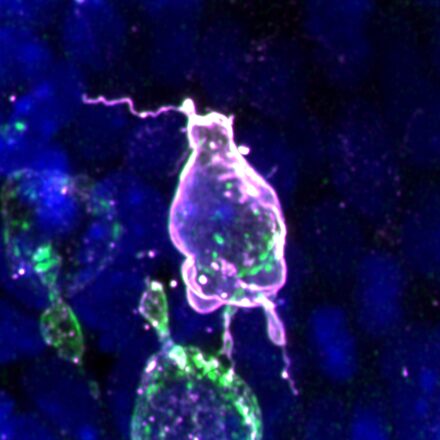
As embryogenesis progresses, intercellular spaces shrink, basement membranes thicken and PGC movement is increasingly restricted. Surprisingly, the group found that when faced with this, the PGC’s nuclear lamina – the mesh-like network of filaments within the inner nuclear membrane – was depleted. This allowed the nucleus to soften, becoming wrinkled and deformed in appearance. Whilst this may aid migration in confined spaces, it also presents an unexpected risk to DNA integrity, and in some cases, leads to nuclear rupture and cell death. Incidences of rupture and DNA damage increased when cellular confinement was increased pharmacologically. This is the first demonstration of confinement-induced DNA damage in a naturally occurring process during embryogenesis, having only been shown previously in cultured cells, and typically only in the context of cancer or immune cells.
Through the comprehensive and dynamic study of PGC migration in mouse embryos, this work offers unprecedented insights into germ cell behaviour and their changing environments during early development. The presence of actin-rich protrusions and ECM proteins underlies the divergence of mammalian PGC migration compared to previously studied models. Interestingly, the impact that increasing confinement has on nuclear stability reveals that PGC migration presents an inherent risk to genome integrity. This raises new questions about how the germline stays genetically stable in such conditions and highlights the potential need for robust DNA repair systems. It also promises new insights into the origins of germline mutations, and widespread conditions such as infertility and germ cell tumours in children, which occur when PCGs fail to reach the gonads.
Finally, this study exemplifies the success of harnessing an interdisciplinary approach to image key fundamental cell biology processes in vivo. Though high-resolution, quantitative analyses can be challenging, by allowing visualisation of cell movement through developing tissue, this approach could be harnessed further to examine the mechanisms behind clinically-relevant cell migration patterns, such as those seen in cancer metastasis, immune cell movement and tissue engineering.
This work was funded by UKRI MRC, the German Research Foundation and the University of Freiburg.
Further references
Primordial germ cells experience increasing physical confinement and DNA damage during migration in the mouse embryo. Goodwin, K., Emrich, E. A., Arnold, S. and McDole, K. Science Advances
Kate’s group page
Sebastian Arnold, University of Freiburg
Related articles
Using ultrasound to determine pregnancy in mice
Animal research statement
As a publicly funded research institute, the LMB is committed to engagement and transparency in all aspects of its research. This research used mice, in accordance with the UK Animals (Scientific Procedures) Act 1986. This work was conducted under a Project Licence, reviewed and approved by the MRC Laboratory of Molecular Biology (LMB) Animal Welfare and Ethical Review Body (AWERB) committee and the UK Home Office.
The LMB uses the minimum number of rodents necessary to achieve results and only uses animals in research where there are no suitable alternatives, in line with the 3R’s (replace, reduce, refine). We currently work with fruit flies, nematode worms, mice, rats and zebrafish.